Tissue regeneration
Research Group Tissue Regeneration
The Research Group Tissue Regeneration, led by PD Dr. Anika Jonitz-Heincke, investigates cellular and molecular mechanisms of bone and cartilage regeneration at the Biomechanics and Implant Technology Research Laboratory (FORBIOMIT) of Rostock University Medical Center. The aim is to derive new strategies for the treatment of joint diseases, bone defects and implant-related conditions from basic science findings. The research group is embedded in several large interdisciplinary research projects.
Cell Biology Expertise and Primary Cell Cultures
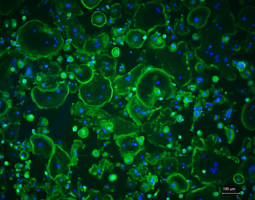
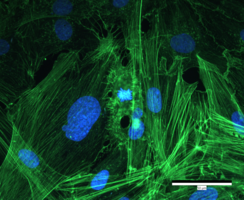
The research group has extensive experience in isolating and culturing human primary cells from sources including femoral heads, hyaline articular cartilage, buffy coats, and whole blood. These cells serve as the experimental basis for all research projects within the group and enable the investigation of biological questions under physiologically relevant conditions. Cultivation is performed in both two-dimensional monolayers and three-dimensional culture systems that more closely mimic the native tissue environment. The established primary cell cultures include osteoblasts, osteocytes, osteoclasts, and chondrocytes, as well as fibroblasts and PBMCs. This broad range of cell types enables the group to address both bone and cartilage regeneration and inflammatory processes at the cellular level.
Electrical Stimulation for Bone and Cartilage Regeneration
A central research focus of the group is the targeted use of electric fields to promote tissue regeneration. This work is embedded in the DFG Collaborative Research Centre 1270 “Electrically Active Implants – ELAINE”.
Endogenous electrical potentials play a pivotal role in the regeneration of natural tissues. The group investigates how alternating electric fields can be harnessed therapeutically to address two major clinical challenges: the repair of cartilage defects and the improvement of implant osseointegration.
To regenerate hyaline cartilage tissue, electrical and mechanical stimulation experiments are conducted using human chondrocytes in monolayer cultures or in hydrogel-based scaffolds. Target parameters include cell morphology, viability, and collagen type I and type II deposition, with the collagen type II-to-type I ratio serving as a measure of redifferentiation capacity. The investigations also take into account sex-specific differences in the cellular response, which may be relevant for the development of personalized therapeutic approaches.
In the field of bone regeneration, the group examines the in vitro effects of alternating electric fields on osteogenic and osteoclastic cells in long-term experiments. Cellular responses vary depending on voltage and frequency, with electrical stimulation substantially promoting osteogenic differentiation and mineralization. The aim is to define therapeutic windows for optimized electrical stimulation of bone regeneration based on these in vitro data.
Material-Induced Inflammatory Reactions
A further key focus of the group is the molecular mechanisms underlying particle-induced inflammatory processes that occur during aseptic loosening of joint endoprostheses. This research is part of the DFG Research Training Group 2901 “Systemic and Local Reactions to Biomaterial Incompatibilities in Joint and Skin Lesions – SYLOBIO”.
Metallic abrasion particles generated by the wear of artificial joints are taken up by bone-forming osteoblasts and trigger a complex inflammatory cascade. The work demonstrates that particle uptake is detectable shortly after initial contact, as evidenced by changes in cell morphology and spectral data, thereby immediately initiating a molecular response. Using time-resolved gene expression analyses, we identify relevant expression profiles of surface receptors and associated intracellular signaling pathways that characterize the interplay between particle exposure and inflammatory activation in osteoblasts. Central to the inflammatory response is the NLRP3 inflammasome – an intracellular protein complex that functions as a sensor of danger signals and regulates the release of pro-inflammatory cytokines such as IL-1β and IL-18. In addition, the influence of released metal ions on the differentiation capacity of human bone cells is investigated. Methodologically, the group employs coculture models that replicate the interactions of different cell types in periprosthetic tissue and generate comprehensive OMIC data, including single-cell analyses, to resolve cell-type- and patient-specific response patterns at the molecular level.
High Hydrostatic Pressure Treatment of Tissue Grafts
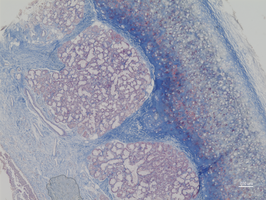
Processing of allogeneic grafts using conventional methods, such as irradiation or chemical decontamination, frequently compromises the biomechanical integrity and inductive properties of the tissue. The group utilizes high hydrostatic pressure technology to reliably devitalize cells in musculoskeletal tissue while preserving the extracellular matrix. In addition to extensive studies on devitalization efficiency, experimental investigations on the revitalization of treated tissues are conducted both in vitro and in vivo. Complementary work focuses on protocols for microbial decontamination and the oncologically safe processing of tumor-infiltrated tissue.
Implant Materials
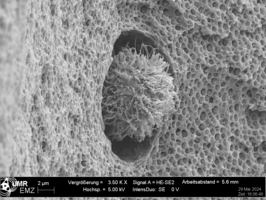
In interdisciplinary collaborations, novel materials for endoprosthetics are biologically characterized, including additively manufactured titanium-niobium-tantalum alloys with bone-like elasticity as well as hybrid materials composed of alumina-toughened zirconia ceramics and titanium alloys, or tricalcium phosphates.


![[Translate to English:] Europäische Fonds](/fileadmin/_processed_/5/a/csm_Fonds_Logo_quer_0d4f666b8f.jpg)
![[Translate to English:] ESF](/fileadmin/_processed_/d/1/csm_ESF_Logo_f_Zusatz_unten_313c60cf7e.jpg)